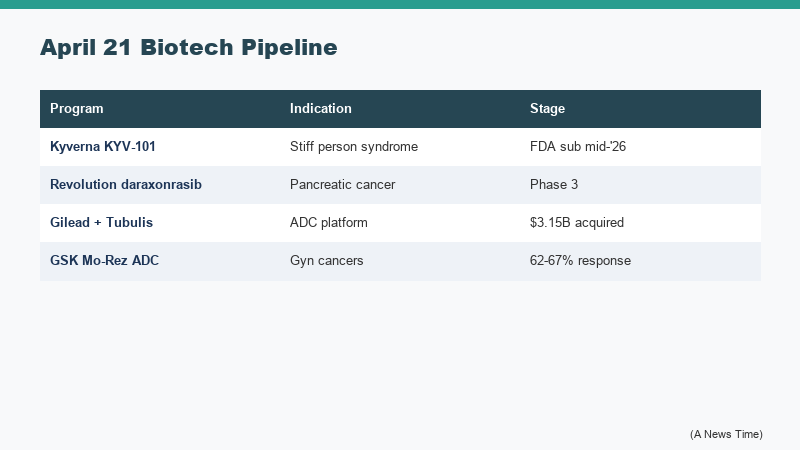
In the space of a single news cycle on , three distinct but thematically linked biotech stories arrived that each, in its own way, extended the frontier of what medicine can actually do. Kyverna Therapeutics disclosed that a personalized CAR-T cell therapy improved mobility and reduced disability in patients with stiff person syndrome, a rare and brutal neurological autoimmune disorder that currently has no approved treatment. Revolution Medicines released Phase 3 data for daraxonrasib, a KRAS inhibitor that nearly doubled median overall survival in metastatic pancreatic cancer compared to chemotherapy alone. And Gilead Sciences announced a $3.15 billion acquisition of Tubulis, a German biotech built around next-generation antibody-drug conjugate technology.
These are not incremental improvements to existing treatments. Each addresses a disease category or tumor biology that the pharmaceutical industry spent decades calling undruggable or treatment-resistant. The science that makes all three possible has matured quietly over years of basic research, and the results reaching clinical visibility now represent the downstream output of investments and failures that predate most of the companies reporting them.
Kyverna's CAR-T for Stiff Person Syndrome: The Biology and What We Know
Stiff person syndrome is a rare, progressive neurological condition caused by the immune system attacking proteins in the brain and spinal cord, particularly GAD65, an enzyme involved in producing the inhibitory neurotransmitter GABA. The result is severe muscle rigidity, painful spasms, and progressive disability. It affects an estimated one in one million people, predominantly women, and has no approved treatment. Current management relies on off-label use of benzodiazepines, baclofen, and immunosuppressants, none of which reliably halt progression.
Kyverna's approach, using its KYV-101 therapy, borrows a mechanism from oncology. In blood cancer treatment, CAR-T cells are engineered from a patient's own T cells to recognize and kill cancer cells expressing a specific surface protein. The same logic applied to autoimmune disease targets B cells, which are responsible for producing the autoreactive antibodies that drive conditions like stiff person syndrome. By depleting B cells, CAR-T therapy effectively performs what researchers describe as an immune system reset: removing the dysfunctional immune population and allowing the immune system to reconstitute from a cleaner state.
"A one-time, personalized cell therapy from Kyverna Therapeutics improved mobility and reduced disabilities in patients with stiff person syndrome. If approved, it would become the first treatment for stiff person syndrome and the first personalized CAR-T therapy for an autoimmune disease of any kind to reach the market."
Adam Feuerstein, Senior Writer, STAT News, April 21, 2026
The clinical results presented Tuesday showed improvements in mobility and disability metrics that are meaningful against a disease baseline where virtually no pharmacological intervention had previously demonstrated reliable efficacy. Kyverna said it intends to submit KYV-101 to the FDA by the middle of 2026. The FDA has previously granted KYV-101 Breakthrough Therapy designation, which provides more intensive FDA guidance and a rolling review process. If approved, it would hold two historical distinctions simultaneously: first approved treatment for stiff person syndrome, and first approved CAR-T therapy for any autoimmune disease.
This second distinction is the one that carries the larger structural implications for the biotech industry. A growing number of companies, including Bristol-Myers Squibb, Novartis, and several smaller biotechs, have been developing CAR-T programs for autoimmune diseases in lupus, myositis, and inflammatory bowel disease. An FDA approval for Kyverna in stiff person syndrome would validate the regulatory pathway and provide a pricing and reimbursement reference point for the entire class. The first approval is always the hardest to get, and the easiest to reference.
Daraxonrasib and the KRAS Mutation: A 40-Year Hunt Finally Delivers
The KRAS gene mutation has a particular status in cancer biology. Identified in 1982, it is the most commonly mutated oncogene in human cancers, present in roughly 30 percent of all tumors and in approximately 90 percent of pancreatic ductal adenocarcinomas, which is the most common form of pancreatic cancer. For four decades, it was considered undruggable: the KRAS protein's smooth, featureless surface appeared to offer no binding site that a small molecule could grab onto with sufficient specificity to be therapeutically useful.
The field cracked open when researchers identified a pocket on the KRAS protein that appeared transiently when the protein was bound to GDP, a finding that eventually led to the first KRAS-specific inhibitor approval in 2021. Revolution Medicines' daraxonrasib takes a different and potentially broader approach: rather than targeting a specific KRAS mutation, it is designed to inhibit multiple KRAS variants simultaneously by binding to a separate region of the protein.
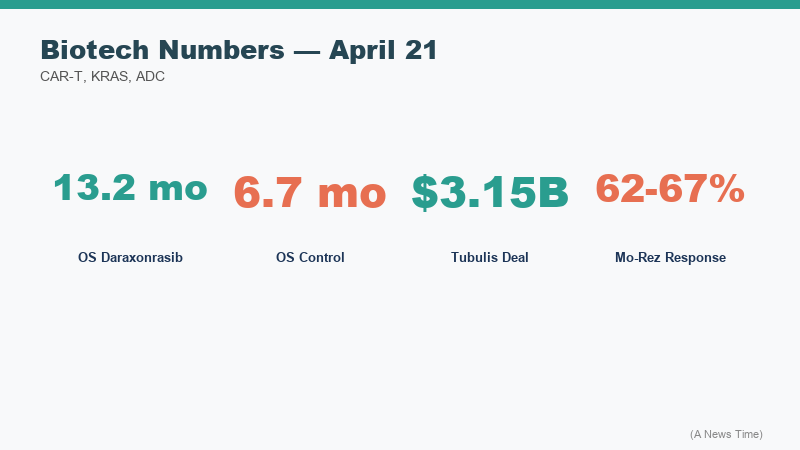
The Phase 3 data Revolution Medicines reported on April 21 showed a median overall survival of 13.2 months for patients on daraxonrasib compared with 6.7 months on standard chemotherapy. That is a near-doubling of median survival in a disease where a six-month median has been essentially unchanged for a generation. The statistic needs context: median overall survival of 13.2 months is not a cure, and metastatic pancreatic cancer remains a disease with very limited long-term survivors. The comparison group receiving chemotherapy had a 6.7-month median that is consistent with existing data on the disease, so the trial's control arm performed as expected.
As the Medicine Maker noted in its reporting on the results, dermatologic toxicities were notable in the daraxonrasib arm, which will influence dosing decisions and combination strategies in future development. Skin toxicities are common across the KRAS inhibitor class and reflect the role of the pathway in normal skin cell proliferation. They are typically manageable but require careful dose titration, and they complicate combination regimens with other agents that have overlapping toxicity profiles.
| Company | Asset / Deal | Stage | Key Data Point |
|---|---|---|---|
| Kyverna Therapeutics | KYV-101 (CAR-T for SPS) | Pre-submission / FDA filing planned mid-2026 | First CAR-T therapy to show benefit in any autoimmune disease; improved mobility and disability scores |
| Revolution Medicines | Daraxonrasib (KRAS inhibitor) | Phase 3 readout | Median OS 13.2 months vs. 6.7 months on chemo in metastatic pancreatic cancer |
| Gilead Sciences | Tubulis acquisition | Deal announced | $3.15B upfront plus up to $1.85B in milestones; adds TUB-040 ADC and tubutecan platform |
| GSK | Mo-Rez ADC | Early clinical | 62-67% tumor response in chemo-resistant ovarian and endometrial cancers |
Revolution Medicines plans to seek regulatory review of daraxonrasib for metastatic pancreatic cancer, though a formal submission timeline was not announced with the Phase 3 data. The company's next steps will likely include discussions with FDA about the clinical meaningfulness of the survival benefit relative to the toxicity profile, and potentially investigating combination approaches with chemotherapy or immunotherapy agents that might extend the survival advantage further.
Gilead and Tubulis: Why ADCs Are Absorbing Billions in Deal Activity
The third major story of the 48-hour period requires understanding what antibody-drug conjugates actually are, because the deal volume around the technology is otherwise difficult to contextualize. An ADC links a cancer-targeting antibody to a cytotoxic drug payload via a chemical linker. The antibody carries the toxic payload through the bloodstream to tumor cells expressing the target protein, where it is internalized and the payload is released inside the cancer cell. The appeal is precision: a cytotoxic agent that would cause unacceptable systemic toxicity at effective concentrations when given alone can be delivered directly to tumor cells at concentrations that kill them while sparing normal tissue.
Gilead's acquisition of Tubulis for $3.15 billion upfront, with up to an additional $1.85 billion in milestone payments, buys it two clinical-stage assets and a platform. The lead asset, TUB-040, targets NaPi2b, a protein expressed on the surface of ovarian cancer and non-small cell lung cancer cells, and is in Phase 1b/2 trials. The second asset, TUB-030, is also in clinical development. Central to the deal is Tubulis' tubutecan linker-payload system, which the company designed to improve tumor selectivity and payload delivery efficiency. Gilead said the Munich facility will operate as a dedicated ADC research center within its structure.
The ADC deal activity in this 48-hour period was not limited to Gilead. GSK reported early clinical data for its own ADC, Mo-Rez, in chemo-resistant ovarian and endometrial cancers, with tumor shrinkage or elimination in 62 to 67 percent of patients in a heavily pretreated population where few options remained. GSK said it plans to move rapidly into late-stage development. The back-to-back ADC disclosures from two large pharmaceutical companies with credible late-stage pipelines suggest the platform has moved past proof-of-concept into a genuine competitive race for market position across multiple tumor types.
For broader context on biotech deal structures and what platforms like Tubulis represent for the oncology pipeline, our earlier coverage of how the top biotech companies have been repositioning after market cap losses heading into 2026 is relevant, as is our reporting on the UK biotech startups building adjacent precision-medicine capabilities. The FDA's recent decision to publish complete response letters in near real time is also worth noting as a transparency shift that will affect how all of these programs navigate the approval process.
What We Still Do Not Know
Each of these stories comes with genuine uncertainty that anniversary coverage and press-release science tend to flatten out.
On Kyverna: the stiff person syndrome data presented April 21 came from a clinical trial in a rare disease with a small patient population. Rare disease trials are structurally constrained in what they can power statistically, which means that the clinical evidence base will be thinner at the time of FDA submission than it would be for a more prevalent indication. The FDA's Breakthrough Therapy designation provides more intensive guidance but does not lower the evidentiary bar; it accelerates the process of reaching an evidentiary decision. The long-term durability of the immune reset, specifically whether B cell depletion followed by reconstitution produces a permanent remission or a temporary one that eventually relapses as B cells return, is genuinely unknown. CAR-T manufacturing is also still a complex, costly, and time-consuming process: a personalized therapy made from a patient's own cells requires weeks of manufacturing per patient, which creates access and scale constraints that a large commercial launch will have to navigate.
On daraxonrasib: the 13.2-month median overall survival is statistically and clinically significant relative to the 6.7-month control, but the absolute number is still a median. Half of patients lived longer; half did not. The dermatologic toxicities noted in the trial were described as "notable" by clinical observers, which is a clinical trial euphemism for "present in enough patients and with enough severity to warrant careful attention in clinical practice." The drug has not yet been through the regulatory review process, and the labeling that emerges from that process, including dose recommendations, contraindications, and risk mitigation strategies, will substantially affect how useful it is in practice.
On the ADC platform broadly: the mechanism is elegant, but the history of oncology is full of mechanistically elegant approaches that showed strong early clinical signals and then ran into durability and resistance problems in later follow-up. The ADC field lost significant momentum in the early 2010s after early clinical disappointments, and while the current generation of conjugation chemistry and linker technology is substantially more sophisticated, the limits of ADC durability at scale across heterogeneous tumor populations are not yet fully mapped.
"RFK Jr. told Congress this week that China is eating our lunch on new drug approvals and clinical trial starts. The irony is that the programs making the most noise right now, from KRAS inhibitors to CAR-T for autoimmune disease, are products of the American biomedical research enterprise that he oversees as HHS Secretary."
Endpoints News, reporting on HHS Secretary Robert F. Kennedy Jr.'s Congressional testimony, April 21, 2026
There is also a policy dimension. HHS Secretary Robert F. Kennedy Jr. told Congress on April 21 that "China is eating our lunch" on new drug approvals and clinical trial starts, per Endpoints News reporting on the hearing. That framing sits in tension with the same 48-hour news cycle showing American biotechs at or near the submission stage in two historically intractable diseases. The FDA's regulatory processes, NIH funding pipelines, and venture capital infrastructure that produced Kyverna and Revolution Medicines are the same institutional stack that Kennedy's budget proposals would reduce. These two facts can coexist; they are worth holding simultaneously.
What to Watch Next
The most consequential near-term event is Kyverna's FDA filing, expected mid-2026. The agency will have to determine whether the clinical data from a rare disease trial in a small patient population is sufficient to support accelerated approval, standard approval, or some conditional approval pathway. Its decision will set the reference for how other CAR-T autoimmune programs are reviewed.
For daraxonrasib, the next question is regulatory strategy: whether Revolution Medicines files for approval on the Phase 3 overall survival data alone, or waits to collect additional follow-up or combination data that might strengthen the label. In pancreatic cancer, where regulatory precedent has been set by drugs with more modest survival benefits, the current data is arguably sufficient for a filing, but the dermatologic toxicity pattern may prompt additional negotiation around risk evaluation and mitigation strategies.
The ADC field, now capitalized with billions in fresh deal money from both Gilead and GSK, will move its programs into late-stage trials over the next two years. The clinical read on TUB-040 in ovarian cancer and non-small cell lung cancer, and on Mo-Rez in gynecologic cancers, will determine whether the technology maintains its promise when exposed to the heterogeneity of real patient populations at Phase 3 scale.
For the biotech sector's structural trajectory, our earlier coverage of China's CRISPR base-editing therapy that freed five beta-thalassemia patients from blood transfusions and of the FDA's accelerated approval of Kresladi for severe LAD-I provides useful context. The regulatory and scientific infrastructure for precision medicine approvals in rare and ultra-rare diseases is more mature now than at any prior point, which is part of what makes this week's Kyverna data actionable rather than merely promising.
A generation ago, stiff person syndrome had no treatment, KRAS was undruggable, and ADC chemistry was too imprecise to deliver consistent clinical benefit. The biological frontier moves slowly until, in a narrow window of time, it moves quickly. This week was one of those windows.
Frequently Asked Questions
What is stiff person syndrome and why has it been so difficult to treat?
Stiff person syndrome is a rare progressive neurological autoimmune disorder in which the immune system attacks glutamic acid decarboxylase (GAD65), an enzyme involved in producing the inhibitory neurotransmitter GABA. This causes severe muscle rigidity, painful spasms, and progressive disability. It affects approximately one in one million people. No treatment has ever been FDA-approved for the condition. Current management relies on off-label use of benzodiazepines, muscle relaxants, and immunosuppressants, which help symptoms but do not reliably halt the disease.
What makes Kyverna's CAR-T therapy different from existing CAR-T cancer treatments?
Existing CAR-T therapies target blood cancer cells expressing specific surface proteins. Kyverna's KYV-101 targets B cells, which are responsible for producing the autoreactive antibodies that drive autoimmune diseases. By depleting B cells, the therapy performs an immune system reset, removing the dysfunctional immune population. The same mechanism is being investigated for lupus, myositis, and other autoimmune conditions by multiple companies. If KYV-101 is approved, it would be the first CAR-T therapy approved for any autoimmune disease.
Why has the KRAS mutation been so hard to target for so long?
The KRAS protein was identified as a common oncogene in 1982 but spent four decades classified as "undruggable" because its surface appeared smooth and featureless, offering no obvious binding site for a small molecule drug. Progress came when researchers identified a transient binding pocket on the GDP-bound form of the protein. Revolution Medicines' daraxonrasib takes a different approach, targeting a separate region of the protein to inhibit multiple KRAS variants simultaneously, which accounts for its efficacy in the roughly 90 percent of pancreatic cancers driven by KRAS mutations.
What is an antibody-drug conjugate and why is it attracting so much deal activity?
An antibody-drug conjugate (ADC) links a cancer-targeting antibody to a cytotoxic drug payload via a chemical linker. The antibody delivers the toxic payload directly to tumor cells expressing the target protein, allowing drugs that would be too toxic at systemic doses to be used effectively at the tumor site. The current generation of ADC technology, with more sophisticated linker chemistry and payload design, has produced more consistent clinical benefits than earlier generations, driving substantial M&A activity including Gilead's $3.15 billion acquisition of Tubulis and GSK's Mo-Rez program.
What are the limitations of the April 2026 biotech results?
Kyverna's stiff person syndrome data comes from a small rare-disease trial; long-term durability of the immune reset is unknown, and manufacturing a personalized therapy from each patient's own cells is complex and costly. Revolution Medicines' daraxonrasib data shows a meaningful but not curative survival benefit, with notable dermatologic side effects. ADC platforms broadly have a history of early clinical promise that has sometimes not translated to sustained benefit at scale, and the current generation, while more sophisticated, has not yet been tested across the full heterogeneity of real patient populations in Phase 3 settings.
Sources
- Kyverna Therapeutics plans to submit cell therapy for stiff person syndrome for FDA approval, STAT News
- This Week's Biopharma News: KRAS Breakthrough and ADC Momentum, The Medicine Maker
- Daraxonrasib Demonstrates Unprecedented Overall Survival Benefit in Metastatic Pancreatic Cancer, Revolution Medicines
- RFK Jr. says China is eating our lunch in biotech advances, Endpoints News













